Removing the Irritation from Ammonia Sampling Systems: In-Situ Water Analysis Anhydrous Ammonia

In-Situ Water Analysis for Anhydrous Ammonia
Ammonia is unfortunately also literally irritating. It is irritating to breathe, it is irritating to the eyes, it is irritating to the skin, and is therefore irritating to take a grab sample of ammonia, a task required at ammonia production facilities for various purposes. One such sampling requirement centers on measuring the very small percentage of water content mandated for (otherwise) anhydrous ammonia. By U.S. Department of Transportation decree, certain grades of anhydrous ammonia product need to contain a minimum of 0.2% water by volume to be transported by truck or rail cars as this water content works to inhibit rapid steel corrosion in transportation vessels. Conversely, the maximum water content permitted for commercial grade anhydrous ammonia is 0.5%, above which it becomes problematically contaminated and/or diluted.
As a critical component to the agricultural industry, it is therefore imperative that ammonia water content be accurately measured so that the product is delivered safely and without corrosion to the existing array of steel transportation infrastructure. If water content in anhydrous ammonia drops to exceedingly low levels, then corrosion may occur rapidly. Ammonia producers must therefore verify that the water content in their product streams is above minimum thresholds for each grade of product prior to custody transfer.
Enter the need to sample. Historically, grab samples of ammonia are taken in the field and carted off to the lab for this analysis. This requires onsite operations and lab manpower and often involves – at a minimum – the risk of exposure. Consequently, full personnel protective equipment (PPE) is typically required in the form of face shield, gloves, suit, and respirator.

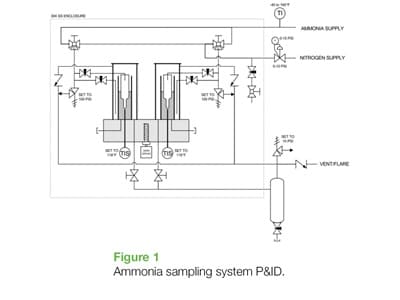
To help make the measurement of all this new ammonia hopefully easier and safer, Swagelok has developed an automatic sample system designed to make the necessary water analysis measurement in-situ without the need to physically take a grab sample off to a lab.
The sampling method follows closely the general requirements set forth as per CGA G-2.2, but improves on a few key points. The ammonia sample collects inside of a larger 250mL residue tube as opposed to 100 mL, which is a larger volume that reduces the error in the procedure. As ammonia will boil at -28°F (14.7 psia), it is challenging to take a representative sample without rapid evaporation, which makes it difficult to ensure precision in the actual volume of the sample. Additionally, rapid evaporation will also inadvertently carry off some of the very water intended for measurement, resulting in a misleadingly low level of residue water.
To combat this challenge, the sample system first cools the outside of the sample residue tube with liquid ammonia in a separate, closed glass annulus. This ammonia bath pre-chills the sample glass and minimizes the evaporation that occurs when the actual ammonia sample is taken inside of the residue tube. Once a 250mL sample has been taken, a 200 W aluminum block heater is activated to slowly and carefully boil off the ammonia so as not to entrain water intended for analysis. This process takes 40 minutes. After the ammonia has been evaporated, a small amount of water will remain inside of the residue tube. The operator may then read the water content directly from the glass residue tube to ensure the desired water content range (usually between 0.2 – 0.5%), without ever having to “suit up” and physically cart off a sample to the lab.
The sample system contains two residue tubes so that samples may be taken in parallel for quality verification purposes. It is also entirely contained within a closed glass fixture connected to a vent system, all of this inside a larger purged steel enclosure. Improvements to the system are also ongoing based on additional feedback from existing installations, mostly to protect the electronics associated with the heater automation, which are prone to attack from ambient ammonia vapors.
Our goal is to keep improving the accuracy and safety of this measurement. We want to make sure that anhydrous ammonia gets where it needs to go, without the irritation.
