Handling Sour Samples: The Journey Towards Zero-Exposure

The Journey Towards Zero-Exposure
One of the recent trends we have observed with rising emphasis is attention to and reduction of H2S personnel protection monitor “hits” at refineries and other facilities processing H2S laden, “sour” media. In 2010, the American Conference of Governmental Industrial Hygienists (ACGIH) – one of a handful of organizations informing benchmarks for jurisdictional agencies such as OSHA – changed the recommended exposure limits for airborne H2S exposure down to a threshold limit value (TLV) of 1 ppm over an 8-hour time-weighted average and a short-term exposure limit (STEL) of 5 ppm. Previously these values were 10 ppm and 15 ppm, respectively. While the enforceable OSHA limits remain slightly higher, many Health, Safety & Environmental (HSE) teams at processing facilities have taken note and are keen to comply with lower thresholds.

Fundamentally, any further mitigation of this risk (other than suiting up with fresh air, which is never popular) must be done by venting / purging the temporary connections (i.e., quick connects or similar) used with the sample cylinder whenever connecting and disconnecting the cylinder in the field and – often – in the lab as well. While certain lab analyses are performed underneath a vacuum hood with sufficient shield protection for lab personnel, there are also many sample cylinder extractions (i.e. – GC injection) that may not always have the luxury of a hood.
Altogether the journey towards a “zero-exposure” world requires additional facilities in the form of inert gas supply for purging and a vent or drain connection for sour process media to flow. This seems straightforward, and there are many ways to do this, but for, say, dozens of sour sample points in a given plant, this task grows into a larger project to manage, particularly across multiple units in a refinery and the lab.
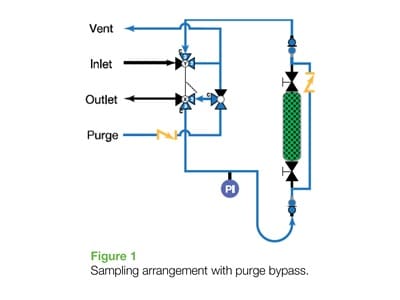
However, this often only addresses half of the problem. The same – or similar – facilities are required in the lab to ensure that the sample cylinder can be safely decoupled and brought back out into the field. Sour media will reside behind the quick connect and build pressure if not vented/purged prior to being sent back out to the field. As the quick-connects see service life and start to leak, this only increases the risk of exposure.

The process of handling sour sample cylinders and maintaining a zero-exposure approach requires a balanced view of the entire process between individual process units, operations teams, lab personnel and the purging/venting resources available at all points. Moreover, a managerial commitment is necessary to enact the necessary changes in order to limit H2S exposures, and we see this more and more. Keeping people safe remains an industry trend, and this is an all-around positive.
